Note: ρ is density, n is refractive index at 589 nm, and η is viscosity, all at 20 ☌ T eq is the equilibrium temperature between two phases: ice/liquid solution for T eq < 0–0.1 ☌ and NaCl/liquid solution for T eq above 0.1 ☌. If Q increases, delta G increases and the reaction becomes less spontaneous. 
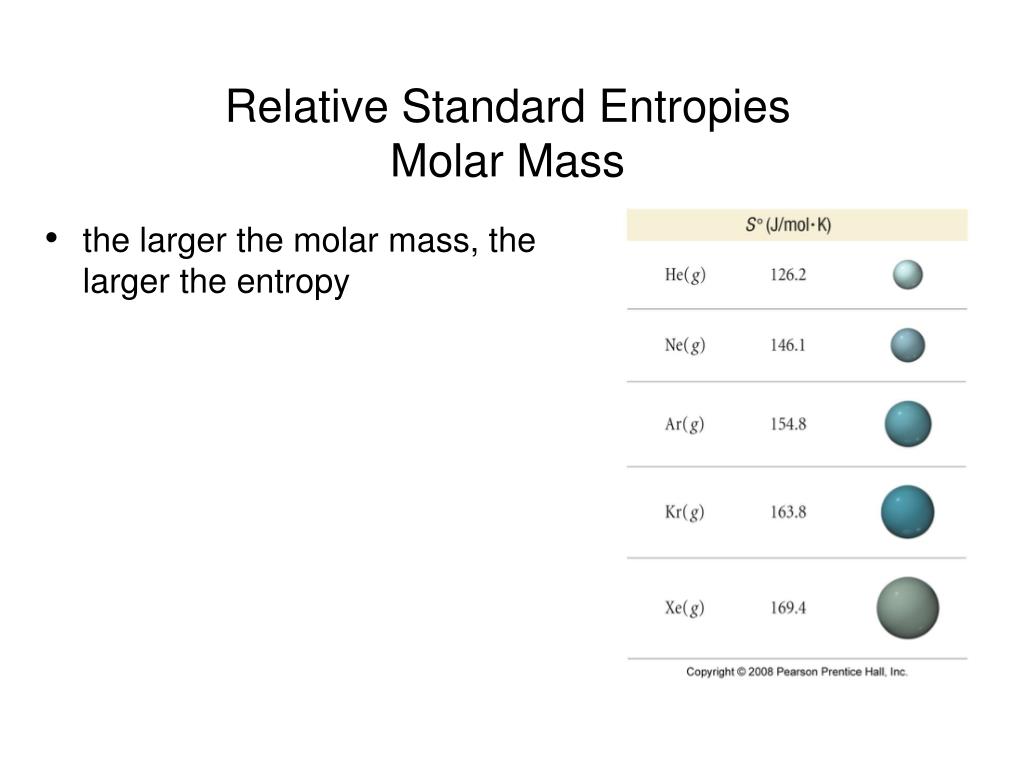
Thermodynamic properties Phase behaviorĭensity data of aqueous solutions Water–NaCl phase diagram Properties of water–NaCl mixtures NaCl, wt% Which one of the following would be expected to have the lowest standard molar entropy C10H22 (s) If delta G is small and positive, the reverse reaction is spontaneous and the system is near equilibrium. However, inclusion of this entire quantity ignores the hydration of the biological structures which reduces. Structure and properties Structure and properties The molar entropy of water at 298.15 K is 69.96. Concept: Entropy is defined as the measure of a systems thermal energy per unit temperature that is unavailable for doing useful work. It is highly recommended that you seek the material safety data sheet ( MSDS) for this chemical from a reliable source such as eChemPortal, and follow its direction. The handling of this chemical may incur notable safety precautions. Mass concentration, g/(100 ml) at 15.This page provides supplementary chemical data on sodium chloride. The standard enthalpy of formation (H0f) of a compound is the change in enthalpy that accompanies the formation of 1 mole of a compound from its elements with all substances in their standard states. the entropy of a pure substance at 298 K and 1 atm pressure). Faraday’s constant, F 96,485 coulombs per mole of electrons. Standard molar entropies are listed for a reference temperature (like 298 K) and 1 atm pressure (i.e. I current (amperes) q charge (coulombs) t time (seconds) Q reaction quotient. Hg (l) Question 2 Which of the following substances would you. Question: Which of the following substances would you expect to have the highest standard molar entropy S° Select one: a. SO, since CF4 is the molecule with largest size and most complex (more particles such as protons, electrons, etc) it has the highest entropy of the molecules given. This problem has been solved Youll get a detailed solution from a subject matter expert that helps you learn core concepts. The more complex the molecule is, the higher the molar entropy. Properties of aqueous ethanol solutions ĭata obtained from Lange 1967 Mass fraction, Standard Molar Entropy, S 0 The entropy of a substance has an absolute value of 0 entropy at 0 K. Re: Determining Increasing Standard Molar Entropy. SO, since CF4 is the molecule with largest size and most complex (more particles such as protons, electrons, etc) it has the highest entropy of the molecules given. Third Law of Thermodynamics (For a Unique Ground State (W1): S -> 0 as T -> 0) and Calculations Using Boltzmann Equation for Entropy Entropy Changes Due to Changes in Volume and Temperature Calculating Standard Reaction Entropies (e.g. The greater the mass of the molecule, the greater will be corresponding constituents. 
The more complex the molecule is, the higher the molar entropy. Molar entropy also depends on the molecular mass of the molecule. 6 Molar specific heats of common gases Principles of Turbomachinery. Uses formula P mm Hg = 10 8.04494 − 1554.3 222.65 + T ĭensity of ethanol at various temperatures Re: Determining Increasing Standard Molar Entropy. 1 Saturated Steam Specic volume, ft 3 / lb Enthalpy, Btu / lb Entropy, Btu / lb. Vapor pressure of liquid P in mm HgĮthanol vapor pressure vs. Molar Entropy is written in joules per kelvin per mole (JK-1mol-1) Solved Examples Determine S for the synthesis of ammonia at 25oc. 
This page provides supplementary chemical data on ethanol.Įxternal MSDS Structure and properties Structure and propertiesĠ.1660 W m −1 K −1 (saturated liquid at 300 K) 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
